OHIO, USA — Earlier this week, the Center for Disease Control and Prevention announced there is a "likely association" between the COVID-19 vaccine and two types of heart inflammation in younger people.
But health officials in Ohio, including Ohio's chief medical officer for the Department of Health,Dr. Bruce Vanderhoff, say most of the cases are mild and harmless and the threat of COVID-19 is far greater.
Dr. Vanderhoff welcomed local experts on Thursday to talk about the possible side effect caused by the MRNA COVID-19 vaccines. Myocarditis and pericarditis are inflammations of the heart muscle and areas around the heart, respectively. Both can cause heart arrhythmia and affect heart function.
"Signs and symptoms of myocarditis include chest pain, fatigue, shortness of breath, arrhythmia," said Dr. Vanderhoff.
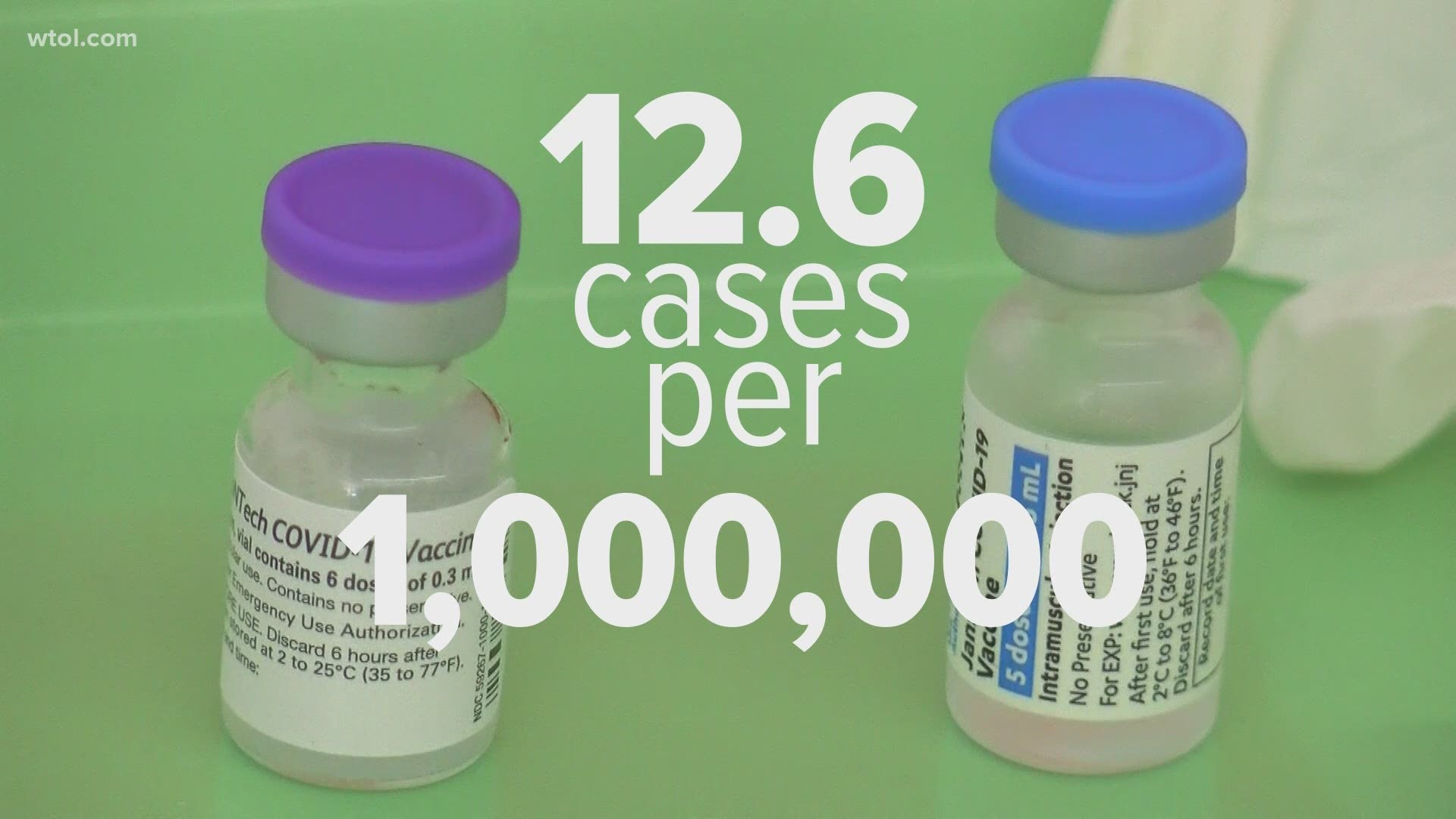
Most of the cases are being found in people 30 years or younger and most of them are male. Still, the cases are extremely rare. There are just 12.6 cases per one million according to the CDC, approximately one one-hundred thousandths of a percent (0.00001%).
Pediatric cardiologist Dr. Simon Lee with Nationwide Children's Hospital in Columbus says the coronavirus poses a bigger threat to the heart than the vaccine.
"Most of these cases appear to be mild and resolve without treatment or significant intervention," said Dr. Lee.
Dr. Lee says myocarditis can be caused by bacterial infections, some everyday medications and even common viruses such as the coronavirus. But he says he hasn't seen any severe cases associated with the vaccine.
And Dr. Vanderhoff says these discussions are proof that they are continually monitoring the vaccine and its long-term effects for safety with systems like VAERS or the Vaccine Adverse Event Reporting System.
We can really have confidence that we have the systems in place to identify this risk, although it is very small," said Dr. Vanderhoff.
He says the FDA and CDC are updating guidance on the shots to include this new but rare condition.
As of June 24, the CDC is continuing to recommend everyone 12 years of age and older gets vaccinated.
RELATED VIDEO: